Knight Therapeutics Inc - ESG Rating & Company Profile powered by AI
Knight Therapeutics Inc - ESG Rating & Company Profile powered by AI
The Disclosure rating covers seventeen UN Sustainable Development Goals including: 'No Poverty', 'Responsible Production & Consumption' and 'Peace, Justice & Strong Institutions'. Full Sustainability assessment of Knight Therapeutics Inc are accessed by registering for free. The Sustainability assessment for Knight Therapeutics Inc represents the company's transparency towards the United Nations SDGs.
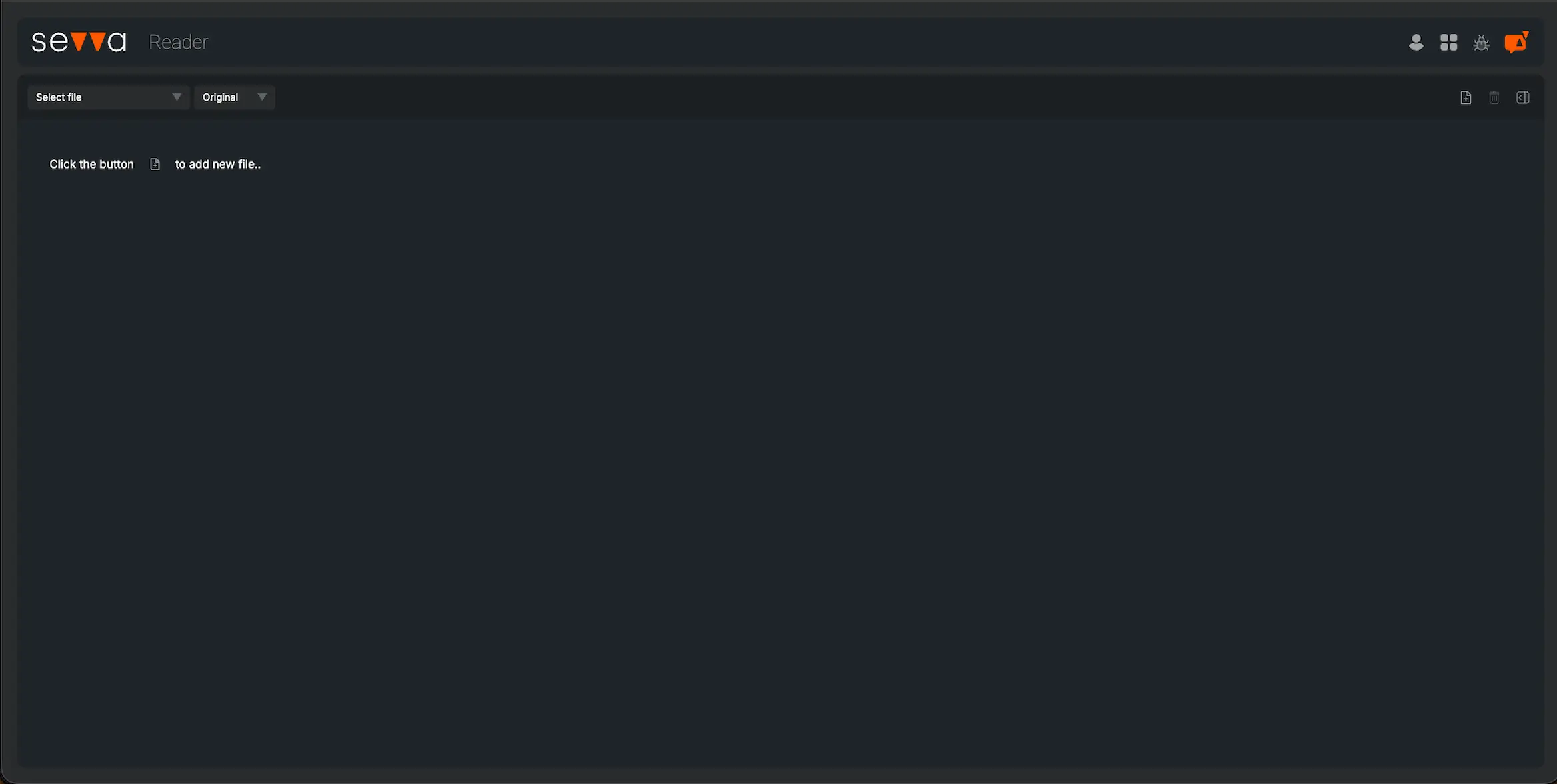
Knight Therapeutics Inc in the Pharmaceuticals industry gained a UN SDG ESG Transparency Score of 5.7; made up of an environmental score of 6.0, social score of 8.0 and governance score of 3.2.
5.7
Medium ImpactEnvironmental
Social
Governance
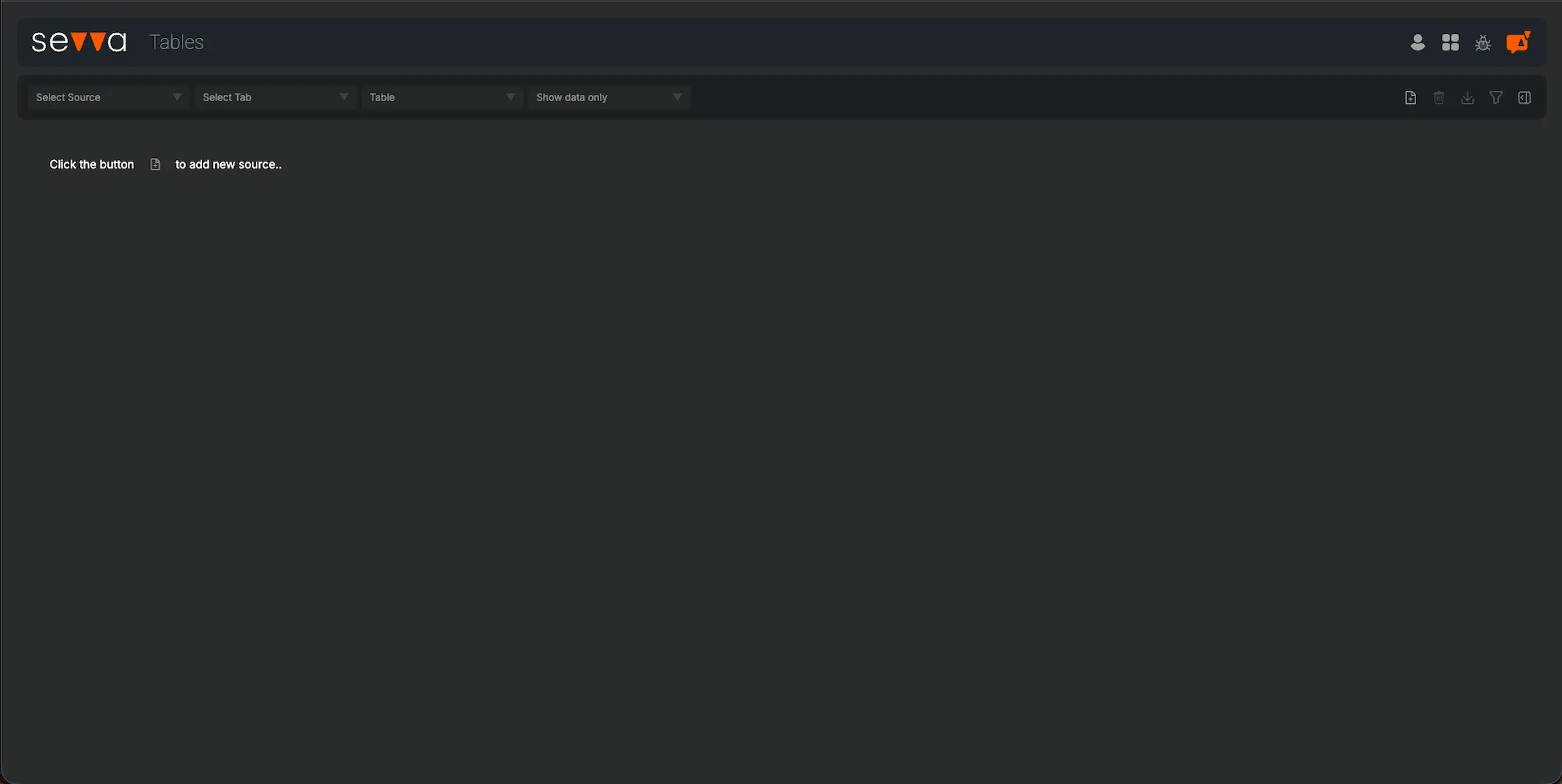
Peer Group Comparison
| Rank | Company | SDG Transparency Score ⓘ | Performance |
|---|---|---|---|
| 1 | Caladrius Biosciences Inc | 8.0 | High |
| 1 | Can Fite Biopharma Ltd | 8.0 | High |
| 541 | Wntresearch AB | 5.8 | High |
| 541 | Zenotech Laboratories Ltd | 5.8 | High |
| 579 | Knight Therapeutics Inc | 5.7 | High |
| 579 | Caplin Point Laboratories Ltd | 5.7 | High |
| 579 | Biotest AG | 5.7 | High |
| ... | ... | ... | |
| 1970 | Zyden Gentec Ltd | 0.0 | Low |
| 1970 | iNtRON Biotechnology Inc | 0.0 | Low |
| ... | ... | ... |
Frequently Asked Questions
Does Knight Therapeutics Inc have an accelerator or VC vehicle to help deliver innovation?
Does Knight Therapeutics Inc disclose current and historical energy intensity?
Does Knight Therapeutics Inc report the average age of the workforce?
Does Knight Therapeutics Inc reference operational or capital allocation in relation to climate change?
Does Knight Therapeutics Inc disclose its ethnicity pay gap?
Does Knight Therapeutics Inc disclose cybersecurity risks?
Does Knight Therapeutics Inc use carbon offsets or credits exclusively for residual emissions (typically less than ~0.5–5% of total emissions)?
Does Knight Therapeutics Inc offer flexible work?
Does Knight Therapeutics Inc have a long term incentive (LTI) executive compensation plan based on a measure of return on capital?
Does Knight Therapeutics Inc disclose the number of employees in R&D functions?
Does Knight Therapeutics Inc plan to change its portfolio composition to lower the emissions intensity of its energy mix (e.g., by shifting from oil to gas, or by adding lower-carbon options like hydrogen, e-fuels, bioenergy, etc.)?
Does Knight Therapeutics Inc conduct supply chain audits?
Does Knight Therapeutics Inc disclose incidents of non-compliance in relation to the health and safety impacts of products and services?
Is there a statment that there is no plan to expand their cement production? (for example: 'We have no current plans to add additional cement making capacity')
Does Knight Therapeutics Inc conduct 360 degree staff reviews?
Does Knight Therapeutics Inc disclose the individual responsible for D&I?
Does Knight Therapeutics Inc disclose current and historical air emissions?
Is there a statment that there is no plan to expand their coal usage? (for example: 'We have no current plans to add additional coal powered electricity generation')
Is executive remuneration linked to climate performance?
Does the Board describe its role in the oversight of climate-related risks and opportunities?
Does Knight Therapeutics Inc disclose current and / or historical scope 2 emissions?
Does Knight Therapeutics Inc disclose water use targets?
Does Knight Therapeutics Inc have careers partnerships with academic institutions?
Did Knight Therapeutics Inc have a product recall in the last two years?
Does Knight Therapeutics Inc disclose incidents of discrimination?
Does Knight Therapeutics Inc allow for Work Councils/Collective Agreements to be formed?
Has Knight Therapeutics Inc issued a profit warning in the past 24 months?
Does Knight Therapeutics Inc disclose parental leave metrics?
Does Knight Therapeutics Inc disclose climate scenario or pathway analysis?
Does Knight Therapeutics Inc disclose current and / or historical scope 1 emissions?
Does Knight Therapeutics Inc explicitly state that carbon offsets or credits are separate from its emissions-reduction progress or that they are not counted toward its emissions-reduction targets?
Are Operating Expesnses linked to emissions reduction?
Does Knight Therapeutics Inc disclose the pay ratio of women to men?
Does Knight Therapeutics Inc support suppliers with sustainability related research and development?
Does Knight Therapeutics Inc disclose the number of operations that have been subject to human rights reviews or human rights impact assessments?
Does Knight Therapeutics Inc reflect climate-related risks in its financial statements?
Is there a statment that there is no plan to expand their carbon intensite energy assets? (for example: 'We have no current plans to carry out further drilling for oil,')
Is Knight Therapeutics Inc involved in embryonic stem cell research?
Does Knight Therapeutics Inc disclose GHG and Air Emissions intensity?
Does Knight Therapeutics Inc disclose its waste policy?
Does Knight Therapeutics Inc report according to TCFD requirements?
Does Knight Therapeutics Inc plan to mitigate emissions from future new production assets through measures such as electrifying equipment, carbon capture and storage, repurposing waste gas, methane leak detection and repair, eliminating flaring, etc.?
Does Knight Therapeutics Inc disclose its policies for bribery, corruption, whistle-blower, conflict of interest?
Does Knight Therapeutics Inc disclose energy use targets?
Does Knight Therapeutics Inc disclose its Renewable Energy targets?
Are emissions metrics verified by STBi?
Does Knight Therapeutics Inc have a policy relating to cyber security?
Potential Risks for Knight Therapeutics Inc
These potential risks are based on the size, segment and geographies of the company.
Knight Therapeutics Inc., a specialty pharmaceutical company, develops, acquires, in-licenses, out-licenses, markets, and distributes pharmaceutical and consumer health products, and medical devices in Canada, Latin America, and internationally. It offers Nerlynx for extended adjuvant breast cancer and metastatic breast cancer; Tafasitamab for relapsed or refractory diffuse large B-cell lymphoma; Pemigatinib to treat metastatic cholangiocarcinoma; Trelstar for advanced prostate cancer and for pain associated with endometriosis; Vidaza for myelodysplastic syndrome; Abraxane for metastatic pancreatic adenocarcinoma; Halaven for metastatic breast cancer and doft tissue sarcoma; and Lenvima for differentiated thyroid cancer, advanced renal cell cancer, and unresectable hepatocellular carcinoma. The company also provides Ladevina for multiple myeloma, myelodysplastic syndrome, mantle cell lymphoma, and follicular lymphoma; Zyvalix for metastatic prostate cancer; Karfib for relapsed or refractory multiple myeloma; Leprid for advanced prostate cancer; Rembre for chronic myeloid leukemia; Ambisome and Cresemba for fungal infection; Impavido for leishmaniasis; Exelon for mild to moderately severe dementia; and Ibsrela for the treatment of irritable bowel syndrome with constipation. In addition, it offers Salofalk for ulcerative colitis; Ursofalk to treat biliary cirrhosis; Imvexxy for moderate to severe dyspareunia; Bijuva for moderate to severe vasomotor symptoms associated with menopause in women with intact uteri; Fibridoner to treat idiopathic pulmonary fibrosis; Toliscrin DPI for pseudomonas aeruginosa lung infection in patients with cystic fibrosis; Toliscrin 1-2 for severe acute or resistant chronic infections; and Tobradosa Haler for chronic lung infections. Further, the company finances other life science companies; and invests in life sciences venture capital funds. Knight Therapeutics Inc. was incorporated in 2013 and is headquartered in Montreal, Canada.